Introduction
Mercury (Hg), Lead (Pb), and Cadmium (Cd) considered as toxic metals. Their presence in the aquatic environment above recommended limits is critical to human health and the ecosystem. The presence of mercury in the aquatic environment and the ecosystem remains an area of research interest because of the toxic nature of mercury to humans. The characteristic of non-biodegradability supports the persistency of mercury ions in the system (Šapanin et al. 2019). Mercury ions released into the environment from ores, industries, and agricultural waste. These ions are transported along the eco-chain into humans resulting in food poisoning, liver, kidney malfunctioning, and carcinogenicity (Mousavi et al. 2011; Alam et al. 2019; Avigliano et al. 2019).
There is a need to reduce the presence of Hg(II) ions from solution. The techniques in current use include membrane filtration, oxidation, sedimentation, distillation, ion exchanger, and adsorption (Abdel-Ghani et al. 2014; Abdolali et al. 2014). All these process and techniques have several setbacks range from high cost, the high volume of waste, and the inability to achieve the limits required by the international community (Páez-Hernández et al. 2005). Mercury ion removal using adsorption is considered an accepted technique since it is simple, less expensive, and environmental friendly (Chen et al. 2019).
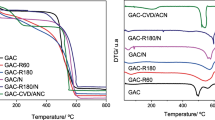
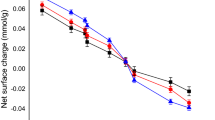
The use of activated carbon as an adsorbent in mercury ion removal exists. The desirability to use activated carbon stems from the fact that activated carbon contains well oriented structural carbon atoms. However, the extra cost of growing bio-materials is problematic, thus readily grown bio-materials such as the African palmae is an added advantage (Thambiliyagodage et al. 2018; Sadegh et al. 2017). The development of activated carbon takes different forms inclusive pyrolysis at high temperature (physical activation) and the use of chemical reagents at low temperature (Jankovi et al. 2019). The use of chemical reagents is preferable since low temperature and shorter time is involved. Besides, the extra surface area added to improve on the reactivity of the activated carbon (Di Stasi et al. 2019). Among the activating reagents, potassium hydroxide is considered the best since it generates high surface area and high porosity of the activated carbon (Yacob et al. 2010; Yang et al. 2019; Wan et al. 2019; Vazquez et al. 2002). The interaction of activated carbon and some mercury ions controlled by the adsorption characteristics of net proton charge and the surface area (Ghaith et al. 2019; Skinner et al. 2007). A Weber-Morris model is an excellent tool in the validation of intra-particle diffusion (Ghannad and Lotfollahi, 2019). The Freundlich isotherm and the extended forms are useful in validating the reaction kinetics of the reaction processes {Yang et al. 2019; Fuadi et al. 2014; Lotfy et al. 2012).
There are several spilt cases of mercury in the environment. Some of these cases could be due to the careless release of mercury from the laboratories, manufacture, and mining operations. In this study, the treatment of mercury in solution with a well prepared and characterized, activated carbon derived from Palmae biomass is done.
Materials and experimental methods
Material preparation
Local communities in Nigeria provide the sampled Palmae shells used as precursor used for the formulation of the activated carbon. The samples were cleaned and air-dried using tap water at a moisture content of 10%. The samples were carbonized and fractionated into powdered and granular forms of 0.6 mm and 1.5 mm, respectively (Ani et al. 2020). The purpose for the fraction is to understand the role of particle fractions in the efficiency of the activated carbons.
Formulation of activated carbon
The carbonization process involved heating of the shells to 4000 °C until the shells turned into a large volume of charcoal. The charcoal was then chemically activated using 75% potassium hydroxide at a moderate temperature of 400 °C for 5 h. The activation was carried out in the ratio of 3:1(carbon: activator) to form a paste. A muffle furnace in an atmosphere of neutral gas (N2) used to heat the paste until dried. The content rinsed with distilled water at ambient temperature. Subsequently, the rinsed and dried content was dried at 105 °C for 24 h to ensure wholistic drying. Again, the content subjected to further pulverization to achieve the PAC-0.6 mm and GAC-1.5 mm fractions. The content ready for adsorption experiments (Bani et al. 2018; Hui and Zaini, 2015). A set of reactions occurring during the activation process is provided:
Characterization of biomass and activated carbon
Proximate analysis
A carbonized shell contains physical and chemical components distributed over the carbon structure. These include the water present in the sample (i.e., moisture content), gases and vapors (i.e., volatile matter), and the fixed carbon contained in the sampled. The moisture content was determined by heating the sample at 105 °C for 2 h in a petri dish and recording the difference in weight before and after heating of the sample. The volatile matter was determined by heating the sample in a crucible at 925 °C for 7 min and recording the difference in weight before and after heating of the sample. The ash content was evaluated by heating the sample at 725 °C for 1 h and recording the difference in weight before and after heating of the sample. The fixed carbon content was evaluated by recording the difference between % of moisture content, volatile matter content, and ash content from 100 (Budianto et al. 2019).
Ultimate analysis (CHNS)
The determination of carbon, hydrogen, nitrogen, and sulfur in biomass CHNS analysis. This Dumas technique centered on the full oxidation of the precursor expressed, in the CHNS components. The % of oxygen-derived by recording the difference between the CHNS and 100 (Fleck 1965). The CHNS Analyzer LabX, Ontario, Canada was used for the ultimate analysis. The reaction of the precursor and excess oxygen releases carbon dioxide, water, nitrogen dioxide, and sulfur dioxide, respectively. A chromatographic column was used for the separation of these components, detected by a thermal conductivity detector (TCD.). The signals from the TCD were related to the concentration of the components of the biomass (Zheng et al. 2019).